We conclude that the observed trends in the total current are directly associated with the loss of oxygen in the metal-oxide lattice and the subsequent dissolution of metallic ions in the electrolyte under anodic polarization. 
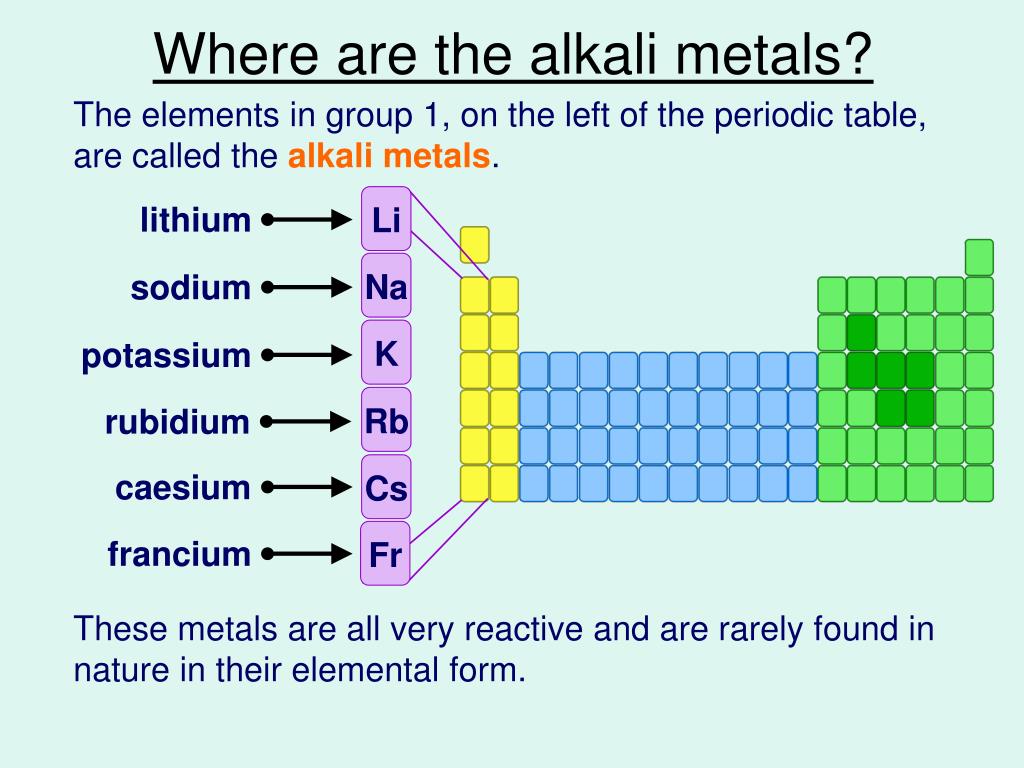
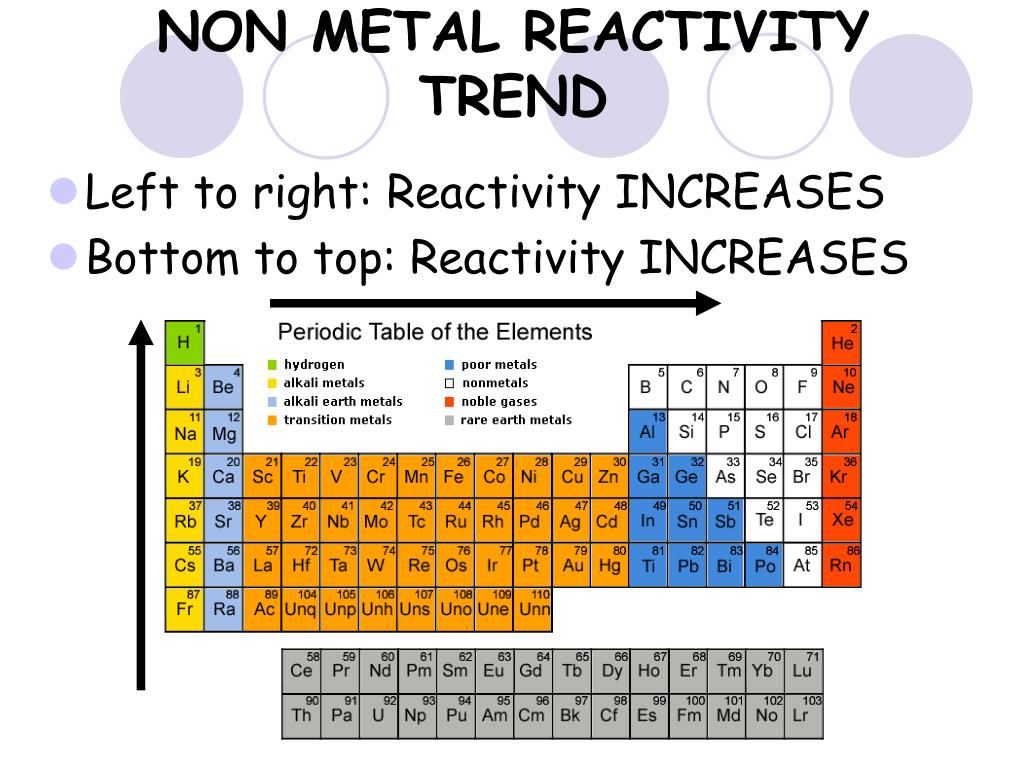
We have shown that basic reactivity trends of pure transition metals can be understood using the NA model on a simple semi-elliptic approximation to the real DOSs. Utilizing small particles increases both the available metal surface area per catalyst metal volume and the number of reactive under-coordinated sites 5. 5a shows a clear trend where the width increases when the center of the distribution shifts up in energy. It is associated with the high potential deposition, which is the origin of the incipient oxidation-corrosion resistance of the lattice. Supported transition metals often serve as catalysts where the metallic part of the catalyst typically is present in the form of nanoparticles on a high surface area support. By operando x-ray absorption, we established that the overall enhancement of the activity for the oxygen evolution reaction is directly attributable to the cross-potential and corrosion process of the electrodeposited materials. The observed activity is strongly related to the lack of stability shown by these composites under anodic polarization revealing a dynamic process ruled by corrosion. It was found that under anodic polarization the materials electrodeposited on gold suffer from a lack of stability, while under cathodic polarization they exhibit stable behavior. A combination of operando x-ray absorption spectroscopy and potentiometric control under aqueous conditions revealed the trends in reactivity yielded by these electrodes, which are directly associated with the cross- and overpotentials as well as the occupancy of the 3d orbitals. The stability of this highest oxidation state decreases from titanium in the +4 state to manganese in the +7 state. We activated gold electrodes for their use as electrocatalyst for water splitting by electrodepositing Cu, Ni and Co. According to the trend, electropositive character of an elements increases as we move from top to bottom in a group of the periodic table. From titanium to manganese the highest oxidation state exhibited, which usually is found only in oxo compounds, fluorides, or chlorides, corresponds to the total number of 3 d and 4 s electrons in the atom. From left to right across a row, the transition metals become increasingly less reactive and more noble in character, following this pattern.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
